Heart Transplant & Circulatory Support
Introduction to Heart Transplant
Heart transplant, or, more exactly, orthotopic allotransplantation of the heart, is nowadays a well-established treatment modality of an end-stage heart failure. First attempts for heart transplant can be tracked down in the beginning of the 20th century when Alexis Carrel carried out the first heterotopic transplantation in a dog. A concept of an orthotopic transplantation in men and its surgical technique was proposed by Norman Shumway in 1960. Using this, the first xenotransplantation of the heart in men (by a chimpanzee heart) was carried out by James Hardy in 1964. It was technically successful, however the donor heart failed due to volume-overload. The first allotransplantation of the heart in men (i.e. using a heart from a human donor) performed Christiaan Barnard in South African Republic in 1967. Nevertheless, these first attempts were stigmatized by unsatisfactory long-term results. The concept of heart transplant was therefore abandoned until introduction of immunosupressives (cyclosporine) into medical practice in 80’s of previous century. From that time on, the expansion of heart transplant has begun once again worldwide, this time, however, with good long-term results.



Currently, the heart transplant is widely accepted treatment modality with satisfactory long-term results regarding mortality, as well as morbidity and quality of life of the patients. More than 5 000 heart transplants are being performed yearly, to summarize, almost 140 000 operations have been performed since 1982. A specific issue of transplantology is a lack of donors and, on the other hand, expanding waiting-list of recipients. Another specific chapter is a demanding long-term patient management after heart transplant, an issue of graft rejection and late side-effects of immunosuppression.
RECIPIENT
A choice of heart transplant recipient is a crucial part of the whole process. An indication for the heart transplant is an end-stage heart failure with severe symptoms and poor prognosis (1-year survival probability of <50%). All other therapeutic modalities are exhausted, beginning with optimal medical therapy, through revascularization procedure, to the implantation of biventricular pacemakers or ICDs. The exact criteria of heart failure severity vary among the transplantation programs of different centres, mostly the include:
- low ejection fraction of the left ventricle (LVEF <20%)
- lowered peak oxygen uptake on exertion (pVO2 max <15 ml/kg/min)
- high pulmonary capillary wedge pressure (PCWP >25 mmHg)
- high plasmatic norepinephrine concentration (>600 pg/ml)
- high level of N-terminal pro BNP (>5000 pg/ml)
- heart arrhythmias
First two characteristics are considered the most reliable predictors of long-term survival. The most common cause of end-stage heart failure is dilated cardiomyopathy, on the second place is the heart failure in coronary artery disease (the two together stand for >90% of indications). Rare indications include heart valve failure (2-3%), congenital heart defects in adulthood (2%), re-transplantations (2-3%) and others.
Every potential recipient must undergo thorough examination with an aim to precisely evaluate the health state and minimize the risk of postoperative complications. The examination comprises routine ECG at rest, 24-Hour ECG Holter monitoring, echocardiography, cardiopulmonary exercise testing (to assess the peak oxygen uptake), right-sided heart catheterization (assessment of pulmonary hypertension), coronarography (in case of coronary artery disease), neuropsychiatric examination and social worker assessment (to evaluate an ability of patient and his family to co-operate on demanding post-transplantation management), biochemical and hematological testing, immunology testing - PRA (panel reactive antibodies to human leukocyte antigens; higher level may point to possible risk of graft rejection), and other specific modalities according to the concrete patient.
Contraindications of the heart transplant. Although there are some exact conditions that may contraindicate the patient for heart transplant, it is always compulsory to individually assess a severity of concrete contraindication and its influence on further prognosis in each specific patient. Here belong:
- active or chronic infection
- other severe comorbidities (malignancy in last 5 years, the period may be shortened in case of definitively treated disease)
- fixed pulmonary hypertension (pulmonary vascular resistance >4 Wood units, transpulmonary gradient >20 mmHg)
- alcohol abuse
- drug abuse
- psychosis
- BMI > 40
Relative:
- age of more than 60-65 years
- diabetes mellitus with late organ complications
- general atherosclerosis
- assumption of non-compliance (not following of treatment instructions, recommendations, misconduct, depression)
- unappropriate social background
- BMI > 30
A recipient must be able to actively cooperate with the transplantation team and be able to early reach the transplantation centre in case of donor finding. Recipients in critical condition require admission to intensive care unit, inotropic pharmacological treatment, in the most severe cases also a mechanical circulatory support in “Bridge to transplant” strategy (See below). These patients are given priority before stable patients on the waiting list in case of donor finding.
DONOR
The legislative of Czech Republic (a law n. 285/2002) determines that the only condition for organ procurement is the identification of brain-death of the potential donor. A previous agreement is not necessary. However, if the patient demonstrably claimed before the death that he does not wish to be an organ donor post-mortem, it is not allowed to proceed an organ procurement. In case the relatives of donor do not agree with it, it is not compulsory to make provision to it. However, it remains an ethical issue.
A potential donor of the heart is every individual with confirmed brain death. Further screening is necessary to exclude severe contraindications for organ procurement and further utilization of the heart. We evaluate age, weight, height, sex, ABO blood group, cause of death, biochemical, hematological and immunological blood testing, chest X-ray, ECG, echocardiography, eventually right-sided heart catheterization and coronarography (in elderly). Contraindications are:
- age of >50-60 years (not an absolute contraindication)
- prolonged cardiac arrest, hypotension, severe chest trauma with a proof of heart injury, intracardial injections
- preexisting heart disease
- extracerebral malignity or glioblastoma
- serological positivity of HIV, HBV; HCV positivity is nowadays not a contraindication anymore
- alcohol or drug abuse - an individual assessment is required
A brain death is associated with an “autonomic and cytokine storm” leading to hemodynamic instability due to autonomic dysfunction, relative hypovolemia due to vasodilation, hypothermia, dysrhythmias. An aggresive volume resuscitation is required together with vasoactive and inotropic support, a hormonal resuscitation (methylprednisone + triiodothyronine + vasopressin). An artificial ventilation, body rewarming and wide-range antibiotic treatment are an obviosity.
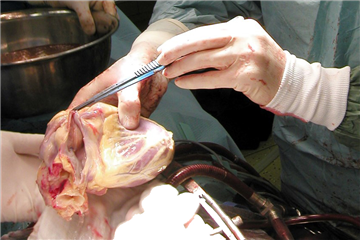
Donor heart procurement. The final and the most important donor and heart assessment is carried out by the cardiac surgical team. After chest opening, the heart is inspected and palpated for evidence of cardiac disease or injury. An anticoagulation is established by a systemic administration of unfractionated heparin (the same as in routine cardiac surgery) and the organ procurement begins. Cardioplegic solution is administered first, inferior caval vein and left heart appendage are incised during the administration to prevent dilation of heart chambers. After finishing the administration of cardioplegia, the inferior caval vein is transected completely. The heart is elevated cephalid and pulmonary veins are divided. Then, the pulmonary trunk, aorta and superior caval vein are divided. The graft is once more checked for eventual pathologies (valve disease, atrial septal defects etc.).

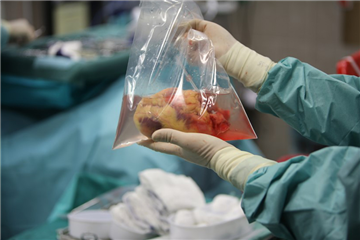
Donor - Recipient Matching. An essential condition is a compatibility in AB0 blood group (an incompatibility may lead to fatal hyperacute rejection). Donor weight should ideally differ in up to 20% from the weight of recipient. Due to various male and female heart weight, we prefer to be lead by "predicted heart mass" index (https://transplanttoolbox.shinyapps.io/calcphm/). Routine prospective performing of other tests (particularly HLA compatibility) is carried out rarely due to time restriction.
Technique of Heart Transplant
A specific sign of heart transplant is the demanding management and coordination of donor heart procurement and transport and simultaneous choosing and preparing of the optimal recipient for the surgery. All this must be done in a rapid fashion in order to not loose ischemic time of the graft. The surgical technique itself has two basic variations. The classical technique of orthotopic heart transplant by Shumway, so called biatrial technique, used to be the gold standard until half of 90’s, and at some centres it has been carried out until nowadays. However, this technique was gradually replaced by a novel bicaval technique that is currently most widespread. The reason for this shift is its lower risk of sinus node injury, better hemodynamic properties and lower risk of tricuspid regurgitation.
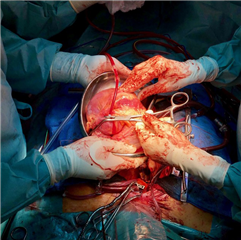
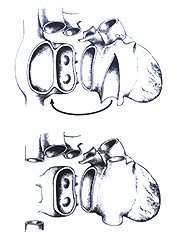 After chest opening and introduction of ECC cannulas (distal placing of aortic cannula, individual cannulating of the caval veins) is the recipient mildly systemically cooled and aorta is cross-clamped. Aorta is divided close to the sinotubular junction, the same for pulmonary trunk. Further process differs according to the chosen technique.
After chest opening and introduction of ECC cannulas (distal placing of aortic cannula, individual cannulating of the caval veins) is the recipient mildly systemically cooled and aorta is cross-clamped. Aorta is divided close to the sinotubular junction, the same for pulmonary trunk. Further process differs according to the chosen technique.
In biatrial technique, both the right and left atrium are transected right above atrioventricular junction, the atrial recipient tissue is preserved in-situ. Left atrium of donor heart is sutured to the left atrium of the recipient by a long continuous stitch. The same works for right atrium. Afterwards, pulmonary and aortic anastomoses are performed.
The bicaval technique is different especially in complete excision of recipient’s right atrium. An island of left atrium with inlets of pulmonary veins is preserved in the same fashion as in biatrial technique. Anastomoses are performed in following order: left atrium, lower caval vein, upper caval vein, pulmonary trunk, and aorta.
Subsequently (in either technique), a thorough de-airing of heart chambers follows, aortic cross-clamp is released and heart begins to beat again after repletion of the blood in coronary vessels. In case of ventricular dysrhythmias, an electrical defibrillation is performed. An extensive inotropic support is often required. The extracorporeal circulation is terminated and the chest is closed.

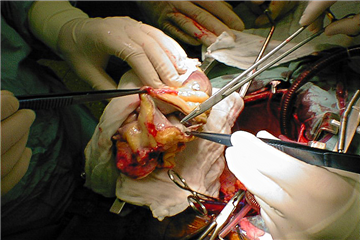
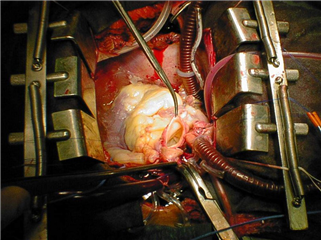

Postoperative Management after Heart Transplant
Allograft often requires prolonged inotropic support in high doses. The donor heart is completely denervated, therefore it does not adequately react to autonomic stress regulation in stress, shock or exertion, it is fully dependent on blood catecholamine levels. Various arrhythmias may occur postoperatively - sinus and junctional bradycardia, atrial fibrillation, ventricular extrasystoles. Eventual systemic, as well as pulmonary, hypertension must be aggressively treated to prevent unnecessary afterload increase. After hemodynamic stabilization, the patient leaves intensive care unit, undergoes the scheduled endomyocardial biopsies and is discharged from the hospital in 3-4 weeks after the surgery. The communication between patient and transplantation centre continues and frequent controls according to the schedule (or in case of complications) are compulsory.
Nenalezeno p8007Graft rejection is a natural reaction of recipient’s immune system to foreign (donor) antigen. Most of the reactions are caused by cellular immune response. Less often, but more severe, is a humoral immune response and subsequent damage to the graft. Hyperacute rejection occurs in the first hours after transplant, is caused by AB0 incompatibility, and is rare. Other forms of acute rejections manifest in the first 6 months after the surgery, their early recognition is possibly only by means of endomyocardial biopsy. Therefore, every patient after heart transplant must repeatedly undergo this examination in a determined schedule. The aim is the early initiation of antirejection therapy in case of rejection. A combination of three drugs has been used as a prophylaxis of rejection: tacrolimus, mycophenolate mofetil, prednisone. If a rejection is detected, further treatment modalities are introduced:
- pulse corticosteroid therapy
- intravenous antithymocyte globuline
- plasmapheresis
- symptomatic management of the patient’s condition (hemodynamic monitoring, inotrope support etc.)
Infections are the most common cause of morbidity and mortality in postoperative period, particularly in the first 3 months after surgery or after episodes of intensive anti-rejection immunosuppressive therapy. To the frequent units belong: pneumonia, mediastinitis and surgical wound infection, urological infections, skin and catheter infections. Apart from standard pathogens (Escherichia coli, Pseudomonas aeruginosa, Staphylococcus species), the predominant agent are Cytomegalovirus, EBV, HSV and other viruses, fungal infections (Candida and Aspergillus), protozoa (Pneumocystis carinii, Toxoplasma gondii).
Nenalezeno p8008Cardiac allograft vasculopathy. A late complication after heart transplant. It is a rapid development of specific form of coronary atherosclerosis in the graft. It is distinguished from “standard” coronary artery disease by a concentric form of disease, diffuse character of lesions, calcifications occur rarely. The main reason of this is probably a combination of immunological reaction (graft rejection) and side-effect of immunosuppressive drugs. The diagnosis is troublesome because of heart denervation - the patients do not fell anginal chest pain, an ideal diagnostic modality is coronarography with IVUS. Treatment options include statins and increased immunosuppressive therapy. The only definitive traetment is re-transplantation.
Malignancies belong to the most severe complications of heart transplant, i.e. of lifelong immunosuppressive therapy. The incidence of malignancies is 10-100 times higher than in general population. The most common neoplasms are lymphoproliferative disorders and carcinoma of the skin. Treatment modalities include immunosuppression dose reduction together with conventional oncological therapeutic approach (chemotherapy, radiotherapy, surgical resection). The procedures are associated with high risk and limited success.
RESULTS
In spite of depicted complications, the heart transplant is currently the most effective treatment modality of end-stage heart failure in comparison to optimal medical treatment and mechanical circulatory support according to the available data. Early postoperative mortality (30 days) is about 5-10%, 1-year survival is 86%.
Short-term mechanical circulatory support
Mechanical circulatory support comprises all the devices used to improve perfusion of the organism, unload heart work and ev. increase coronary perfusion in various clinical scenarios. It is appropriate to divide them into short- and long-term circulatory support due to different indications, techniques and demandingness of application, and most of all, due to different durability and functionality.
Short-term mechanical circulatory support is dedicated to a rapid restitution of organ perfusion. The introduction is relatively fast, often percutaneous. In some devices, a surgical preparation of the vessel, eventually sternotomy a central cannulation are required. It is therefore used in acute and life-threatening conditions. Indications for introduction are:
- prophylaxis (high risk interventional procedures - PCI, TAVI)
- cardiogenic shock (after myocardial infarction and its mechanical complications, in myocarditis and post-partum cardiomyopathy)
- acutely decompensated heart failure
- cardiac arrest and pharmacoresistant ventricular dysrhythmias
- post-cardiotomy low cardiac output (unsuccessful weaning from ECC)
Mechanical circulatory support itself is not a therapy of the cause of hemodynamic instability. It may be understood as a bridging for the heart to convalescence or for further clarification of the pathological condition. Strategies, in which the short-term mechanical circulatory support may be used, are following:
- Bridge to Decision. Maximum 2-3 days after initiation of the support, a re-evaluation is necessary and further strategy must be determined (Bridge to Recovery/Bridge/Transplant). Eventual termination of support in infaust prognosis should be considered, too. Further terms are being used:
- Bridge to Recovery of heart function, without the need of further mechanical circulatory support.
- Bridge to Bridge - a bridging to implantation of a long-term mechanical circulatory support
- Bridge to Transplant - rarely may heart transplant follow after short-term mechanical circulatory support
Even though these methods are capable of significant improvement of patient’s hemodynamic condition, their clear benefit in patient’s survival has been proved in the post-cardiotomy shock only. In other indications, the results are unconvincing, the survival is unsatisfactory and guidelines are not unified. Individual types of short-term mechanical circulatory support are discussed below.
INTRA-AORTIC BALLOON PUMP
 A balloon is inserted to the descending aorta percutaneously through a femoral artery. Synchronously with R-wave of continuous ECG monitoring, it inflates in diastole and deflates in systole. By the inflation in diastole, the perfusion of coronary and supraaortic vessels is being improved (so called diastolic augmentation). In systole, an active deflation leads to rapid reduction of afterload what lowers cardiac workload, and subsequently its oxygen consumption and wall stress. Anticoagulation by heparin is an obviosity.
A balloon is inserted to the descending aorta percutaneously through a femoral artery. Synchronously with R-wave of continuous ECG monitoring, it inflates in diastole and deflates in systole. By the inflation in diastole, the perfusion of coronary and supraaortic vessels is being improved (so called diastolic augmentation). In systole, an active deflation leads to rapid reduction of afterload what lowers cardiac workload, and subsequently its oxygen consumption and wall stress. Anticoagulation by heparin is an obviosity.
The device is mostly used in uncontrollable myocardial ischemia, in cardiogenic shock and in post-cardiotomy low cardiac output. Contraindications are severe aortic regurgitation and aortic dissection. The introduction of device is risky in patients with excess aortic calcifications. To complications belong peripheral limb ischemia, infection, local puncture site complications (bleeding, pseudoaneurysm, fistula, etc.).
EXTRACORPOREAL LIFE SUPPORT (ECLS)
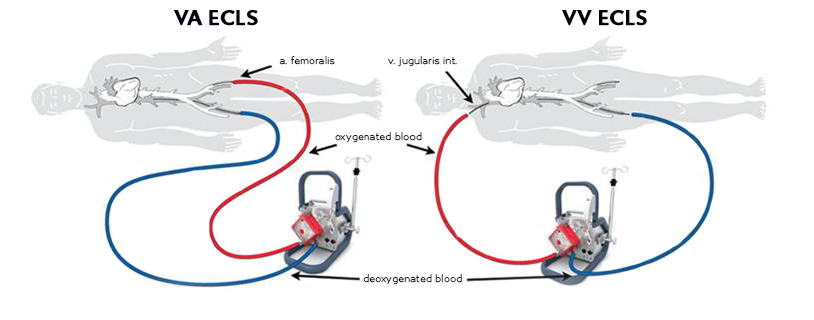
Extracorporeal life support, or the former name - extracorporeal membrane oxygenation (ECMO), is a true mechanical circulatory support in contrast with intra-aortic balloon pump (this only improves perfusion of specified organs). ECLS can be understood as a variation of extracorporeal circulation designed for longer-time use (days to weeks). Thanks to a lack of blood stasis (there is no reservoir) and introduction of heparin-coated cannulas, the anticoagulation does not have to be that intensive as in routine extracorporeal circulation. Components of the device are:
- inflow cannula
- oxygenator with heat exchanger
- centrifugal pump
- outflow cannula
A routine veno-arterial ECLS is introduced through femoral vessels (femoral artery and vein) in a percutaneous or surgical fashion. A central introduction is possible (into the right atrium and ascending aorta) but it is justified only in severe atherosclerosis of peripheral vessels or in post-cardiotomy shock. Another option is axillary artery.
To ensure a safe peripheral limb perfusion, a side cannula for distal perfusion may be of certain benefit. The most common complication is bleeding, as a consequence of necessary anticoagulation. Regarding other complications, an acute respiratory or renal failure, septicaemia, mediastinitis, stroke, oxygenator or pump failure may occur.
Except from the routine veno-arterial setup, there is also a veno-venous setup of ECLS (using jugular and femoral vein) that is not used to support circulation, only to improve oxygenation in respiratory failure. In this case, a former name of ECMO is justified.
TandemHeart
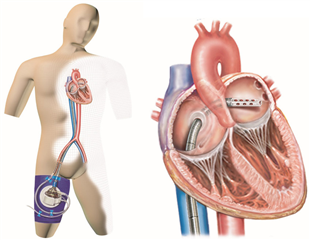 A form of support that is insterted percutaneously through femoral vessels. An inflow cannula passes through the femoral vein and right atrium, then in a transseptal way into the left atrium. It derives blood from there, by which means it unloads the left ventricle. The centrifugal pump itself is placed in a paracorporeal position. The outlet cannula is insterted into the femoral artery. In comparison with ECLS, this device provides better unloading of the left ventricle. It is mostly used in high-risk coronary interventions or in cardiogenic shock after myocardial infarction.
A form of support that is insterted percutaneously through femoral vessels. An inflow cannula passes through the femoral vein and right atrium, then in a transseptal way into the left atrium. It derives blood from there, by which means it unloads the left ventricle. The centrifugal pump itself is placed in a paracorporeal position. The outlet cannula is insterted into the femoral artery. In comparison with ECLS, this device provides better unloading of the left ventricle. It is mostly used in high-risk coronary interventions or in cardiogenic shock after myocardial infarction.
Impella
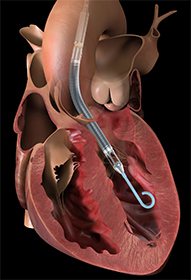 Impella is a technically different type of circulatory support. It is a microaxial pump, that is inserted across the aortic valve into the left ventricle in a retrograde fashion. It provides excellent unloading of the left ventricle, it reduces the heart workload and improves coronary perfusion. It is used mostly as a prophylaxis in catheter interventions where it has proven advantageous over balloon pump. Another indication of use is a cardiogenic shock due to severe left ventricular dysfunction.
Impella is a technically different type of circulatory support. It is a microaxial pump, that is inserted across the aortic valve into the left ventricle in a retrograde fashion. It provides excellent unloading of the left ventricle, it reduces the heart workload and improves coronary perfusion. It is used mostly as a prophylaxis in catheter interventions where it has proven advantageous over balloon pump. Another indication of use is a cardiogenic shock due to severe left ventricular dysfunction.
Levitronix CentriMag
 It is a centrifugal pump with fully magnetically levitated rotation. It is introduced surgically, in a central fashion through sternotomy, it may be designated as either right-sided or left-sided heart support. According to that, the setup is either right atrium → pulmonary trunk or left atrium → aorta. The cannulas are then passed percutaneously to the body surface where the paracorporeal pump is placed. It is used in cardiogenic shock (e.g. in fulminant myocarditis) or in post-cardiotomy low cardiac output syndrome. It has been used with favourable results in the “Bridge to Recovery” or “Bridge to Bridge” strategy. Due to longer durability and functionality (even more than 1 month), it is sometimes referred to as an intermediate-term mechanical circulatory support.
It is a centrifugal pump with fully magnetically levitated rotation. It is introduced surgically, in a central fashion through sternotomy, it may be designated as either right-sided or left-sided heart support. According to that, the setup is either right atrium → pulmonary trunk or left atrium → aorta. The cannulas are then passed percutaneously to the body surface where the paracorporeal pump is placed. It is used in cardiogenic shock (e.g. in fulminant myocarditis) or in post-cardiotomy low cardiac output syndrome. It has been used with favourable results in the “Bridge to Recovery” or “Bridge to Bridge” strategy. Due to longer durability and functionality (even more than 1 month), it is sometimes referred to as an intermediate-term mechanical circulatory support.
Long-term mechanical circulatory support
Long-term mechanical circulatory support aims to improve organ perfusion, unload the left ventricle and eventually increase coronary perfusion, analogous to short-term support. Nevertheless, these devices are technically demanding to install, they require an individual cardiac surgery procedure. On the other hand, they provide a long-term circulatory support, months to even years. They may be set up as a left ventricular assist device (LVAD), right ventricular assist device (RVAD), or biventricular assist device (BiVAD). A total artificial heart implantation (TAH) must be distinguished from the previous devices while it is not only functional but also anatomical replacement of heart.
 Technical solutions. LVAD is implanted with an inflow cannula to the apex of the left ventricle, the pump itself follows (it may be intra- or paracorporeal depending on the chosen device), and the outflow cannula is inserted to the ascending aorta. RVAD derives blood from the right atrium through the pump to the pulmonary trunk. BiVAD is a combination of the both methods. In TAH, it is necessary to remove the native myocardium of ventricles and the device is sutured to the remaining atrial myocardium. Therefore, it is not exactly support but rather replacement of heart function by a device.
Technical solutions. LVAD is implanted with an inflow cannula to the apex of the left ventricle, the pump itself follows (it may be intra- or paracorporeal depending on the chosen device), and the outflow cannula is inserted to the ascending aorta. RVAD derives blood from the right atrium through the pump to the pulmonary trunk. BiVAD is a combination of the both methods. In TAH, it is necessary to remove the native myocardium of ventricles and the device is sutured to the remaining atrial myocardium. Therefore, it is not exactly support but rather replacement of heart function by a device.
Indications for introduction of long-term mechanical circulatory support are similar to heart transplant - end-stage heart failure non-responding to maximum conservative treatment. A specific classification system was designed to distinguish among various clinical profiles of patients with severe heart failure - INTERMACS. However, it is beyond the capacity of this course. On principle, patients are indicated for the implantation when they do not respond to optimal medical treatment, require inotropic support or introduction of short-term mechanical circulatory support, ejection fraction of the left ventricle is below 25%, peak oxygen uptake is below 14 ml/kg/min.
CAVE! Acute cardiogenic shock or acute decompensation of heart failure are indications for introduction of short-term mechanical circulatory support. Long-term support is implanted, if possible, in an elective fashion.
Contraindications for implantation of long-term mechanical circulatory support are irreversible organ dysfunction, severe hemodynamic instability (the introduction of the short-term support is indicated in the case), coagulopathy, complex congenital anomalies, restrictive cardiomyopathy (reduced size of heart chambers), active infection and septicaemia.
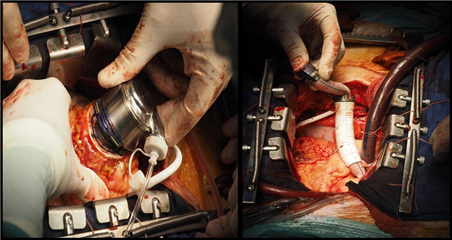
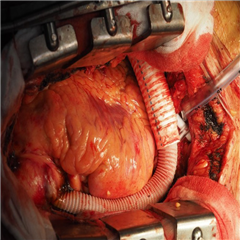

Implantation strategies of long-term mechanical circulatory support are:
- Bridge to Transplant
- Destination Therapy
Bridge to Transplant was one of the essential motivations for a development of long-term support. In patients indicated for heart transplant, the aim is to bridge the period until the heart donor is found. It has been proved that introduction of mechanical circulatory support significantly improves survival of patients with end-stage heart failure before transplant in comparison with optimal medical treatment alone. Simultaneously, the post-transplantation survival is not negatively influenced by the implantation of mechanical support, rather positively.
Destination Therapy is an alternative treatment modality for the patients with end-stage heart failure who are contraindicated for heart transplant (e.g. fixed pulmonary hypertension, extreme obesity). Long-term support is implanted for life. Analogously, it has been proved that implantation of mechanical circulatory support significantly improves survival of these patients in comparison with conservative therapy alone. This strategy has therefore had a rising popularity in last years. Recent studies show that this strategy may be nowadays chosen more frequently than Bridge to Transplant itself.
Fluctuation of the strategy. It is to mention that even though the strategy is exactly defined in the beginning, it is possible that it changes in further course. An extremely obese patient, who was “doomed” for lifelong mechanical circulatory support, may be re-considered and may undergo heart transplant. Sometimes this can be referred to as “Bridge to Candidacy” in case one considers this option from the very beginning. A rare, but possible, reconvalescence from end-stage heart failure with sufficient regaining of heart function may lead to termination of circulatory support (i.e. Bridge to Recovery).
SELECTION OF DEVICE
Technologies of long-term mechanical circulatory support have undergone a heavy technical improvement that has lead to dramatic reduction of morbidity and mortality of the patients. We distinguish three generations of long-term support:
- generation: pulsatile flow pumps (HeartMate I, Thoratec Paracorporeal VAD, Novacor, Berlin Heart ExCor). To preserve pulsatile blood flow, these robust and noisy pumps were manufactured with a need of paracorporeal placement, higher intensity of deterioration and complication risk. Currently, only Berlin Heart ExCor is being produced. It is used in pediatric population.
- generation: axial pumps with continuous flow (HeartMate II, Jarvic 2000, Berlin Heart InCor). Due to markedly simpler construction without the need of many components, these pumps are more compact, less prone to deterioration, easier to implant and the intracorporeal placement is possible (except of the drive line). No negative impact of continuous flow has been observed.
- generation: centrifugal magnetically levitated pumps with continuous flow (HeartWare, HeartMate 3). They have the most compact construction allowing intrapericardial implantation. HeartWare has comparable results to 2nd generation. HeartMate 3 imitates pulsatile blood flow by means of rapid rotation velocity changes, it shows lower long-term morbidity in contrast with previous generation. Mortality remains comparable. Currently, it seems to be the most promising technical solution.
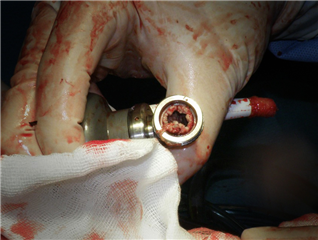
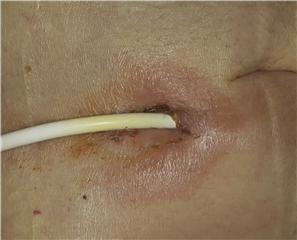 Results of long-term mechanical circulatory support implantation are very satisfactory. Recent data report 1-year survival of 85%, particularly with the newest devices with continuous blood flow. To the complications of long-term mechanical circulatory support and consequent need of anticoagulation belong:
Results of long-term mechanical circulatory support implantation are very satisfactory. Recent data report 1-year survival of 85%, particularly with the newest devices with continuous blood flow. To the complications of long-term mechanical circulatory support and consequent need of anticoagulation belong:
- bleeding (early postoperative, late is mostly gastrointestinal)
- pump thrombosis (the pump requires replacement or systemic thrombolysis)
- right heart failure in implanted LVAD (the afterload of the right ventricle lowers but the preload rises; the condition requires pharmacological treatment, implantation of right-sided support or heart transplant)
- drive line or pump infection (antibiotic treatment, sometimes even pump replacement)
- neurological dysfunction
- renal failure
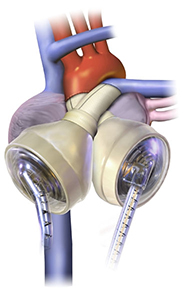 Total artificial heart (TAH) is another modality reserved for some patients (inoperable congenital anomalies or heart rupture according to the newest guidelines). The only device on market is SynCardia TAH. It is a combination of two pneumatic pumps producing pulsatile blood flow. Similarly to the other pulsatile devices, it is relatively robust - one of the important limitations of implantation is a sufficient space in chest for the device. It is used as a bridge to transplant or as a destination therapy in bilateral heart failure. More than 2 000 TAH’s have been implanted until now. The results are relatively satisfying, 72% of patients last out until transplant. Median survival is 554 days.
Total artificial heart (TAH) is another modality reserved for some patients (inoperable congenital anomalies or heart rupture according to the newest guidelines). The only device on market is SynCardia TAH. It is a combination of two pneumatic pumps producing pulsatile blood flow. Similarly to the other pulsatile devices, it is relatively robust - one of the important limitations of implantation is a sufficient space in chest for the device. It is used as a bridge to transplant or as a destination therapy in bilateral heart failure. More than 2 000 TAH’s have been implanted until now. The results are relatively satisfying, 72% of patients last out until transplant. Median survival is 554 days.
Another option is the new biocompatible TAH CARMAT. Its implantation is currently in a phase of clinical testing.
Resources:
- Cohn LH, Adam DH. Cardiac Surgery in the Adult, Fifth Edition. New York: McGraw Hill Education, 2016. ISBN 978-0-07184487-1.
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal 2016; 37: 2129-2200. doi:10:1093/eurheartj/ehw128.
- Schramm R, Morshuis M, Schoenbrodt M, Boergermann J, Hakim-Meibodi K, Hata M et al. Current perspectives on mechanical circulatory support. European Journal of Cardio-Thoracic Surgery 2019; 0: 1-7. doi:10.1093/ejcts/ezy444.
- Málek I. Péče o nemocné po transplantaci srdce. Intervenční a akutní kardiologie 2006; 5: 116-121.
- Ayub-Ferreira SM. Executive Summary - Guidelines for Mechanical Circulatory Support of the Brazilian Society of Cardiology. Arquivos Brasileiros de Cardiologia 2018; 111(1)4-12.
- Schäfers HJ. Klinische Grundlagen der Herz- und Throaxchirurgie, 4., aktualisierte und ergänzte Auflage. Berlin: ABW Wissenschaftsverlag GmbH, 2011. ISBN: 978-3-940615-21-3.
- Napp LC, Kühn C, Hoeper MM, Vogel-Claussen J, Haverich A, Schäfer A et al. Cannulation strategies for percutaneous extracorporeal membrane oxygenation in adults. Clinical Research in Cardiology 2016; 105: 283-296. doi:10.1007/s00392-015-0941-1.
- Birks EJ. Intermediate- and long-term mechanical circulatory support. UpToDate 2019. https://www.uptodate.com/contents/intermediate-and-long-term-mechanical-circulatory-support
- Jeevanandam V, Eisen HJ, Pinto DS. Short-term mechanical circulatory assist devices. UpToDate 2019. https://www.uptodate.com/contents/short-term-mechanical-circulatory-assist-devices
- United Network for Organ Sharing. https://unos.org/data/transplant-trends/
We hereby thank Michal Smetana, M.D., Ph.D. (who worked in Institute for Clinical and Experimental Medicine in Prague) for the practical consultation regarding heart transplant and application of mechanical circulatory support.
A special thanks belongs also to Assoc.Prof. Ondřej Szarszoi, M.D., Ph.D, a vice-chief of Cardiovascular Centre in Institute for Clinical and Experimental Medicine in Prague, for providing the photographic illustrations and further practical consultation of the text correctness.
