Extra-Corporeal Circulation
DEFINITION
Extracorporeal circulation (ECC) is a device, whose primary function is to replace blood circulation (perfusion) a respiration (oxygenation) during heart arrest in surgery of heart and great vessels. It is able to regulate body temperature, as well. In specific occasions it is possible to install additional functions, such as haemoconcentration or haemodialysis.
Successful using of ECC is based on knowledge and application of physical principles and on understanding of physiological and pathophysiological processes of human organism. The aim is to achieve balance between physical and chemical processes (mechanics, rheology, thermics, acid-base balance, etc.), preserving physiological functions (perfusion, gas exchange), pharmacological influence (heparin, protamine, catecholamines, vasodilators) and biological response of the body to this specific situation (systemic inflammatory reaction - SIRS).


Principles of ECC
PERFUSION
Adequate perfusion of patient’s body during ECC can be achieved only by maintaining adequate pump flow and perfusion pressure. The pump flow height is based on body size, age, actual body temperature and other factors. It can be calculated for individual patient using this equation:
| |
l . min-1 . m-2 |
| Normothermia |
2,5 - 2,8 |
| Slight hypothermia |
2,2 - 2,4 |
| Under 28 °C |
1,6 |
| Under 20 °C |
1,2 |

Nevertheless, it is recommended to adjust pump flow during ECC to the general condition of the patient. For instance in elderly patients or patients with arterial hypertension the output should be increased to about 3 l . min-1 . m-2 .
In insufficient pump flow there is a decrease in systemic oxygen consumption (VO2) and the entire capillary bed is not well perfused, which results in lactate increment and severe metabolic acidosis.
In high pump flow there is a risk of traumatisation of blood elements in the ECC system (high pressure gradient, rotations of the pump). Blood is increasingly exposed to artificial surfaces and there is a higher chance of gas embolization (cavitation and bubbles in the circulating blood).
Recommended values of perfusion pressures during ECC vary between 50- 75 mmHg, while maintaining the required calculated pump flow. In high perfusion pressure there is a danger of disconnection of the ECC tubing system and of bleeding complications. In low pressure there were some concerns of brain damage, however, this does not occur if adequate flow is maintained. The excerptions are risky patient groups (edlerly patients with severe aortic atherosclerosis, hypertension and diabetes).
The blood flow in the arterial line is non-pulsatile. Some technical devices enable pulsatile blood flow - it has proven advantages with regard to microcirculation (additional energy for the transport of erythrocytes in the capillaries), in clinical practise, however, there is not any proven difference in mortality and morbidity.

GAS EXCHANGE
During heart arrest, the circulation in the lung system is interrupted and either is the gas exchange in blood. The partial pressures of O2 and CO2 in the desaturated venous blood must be corrected to arterial levels before returning to the systemic circulation. The changes in pO2 and pCO2 are done in oxygenator depending on the controlled flow of mixed gases (air and O2). Oxygen difuses into the blood plasma across a membrane on the basis of gradient between high pO2 in the device and low pO2 in the venous blood flowing from the patient. The principle of CO2 exchange is exactly the opposite. The difference is that this process is always more efective than the process of oxygenation (because of the high degree of diffusion of CO2 in plasma, even if the pressure gradient is not that great).
Once extracorporeal circulation is started, quality of oxygenation must be constantly monitored on the basis of acid-base balance. The value of pO2 in the arterial blood is an indicator of the oxygenator function and the value of O2 saturation in the mixed venous blood from the patient is an indicator of sufficient tissue oxygenation. The value of arterial pO2 should be between 85-200 mmHg (11,3 – 26,6kPa)- below this value the body is in danger of hypoxia, above this there is a risk of oxygen toxicity, formation of microbubbles and free oxygen radicals.
REGULATION OF TEMPERATURE
While the ECC is in function, the temperature of pumped blood can be actively manipulated and thus, the temperature of the patient. In cardiac surgery, this method is often used to induce hypothermia during the surgery and to restore normothermia in the end of it.
Hypothermia slows down the cell metabolism and thus decreases tissue demand of oxygen. During ischemic periods, the available stored energy is capable of maintaining cell viability. Therefore, in the reperfusion phase it is possible to restore normal tissue structure and function. Thanks to systemic hypothermia we are able to decrease blood flow during ECC - it leads to lower traumatization of blood elements and better protection of myocardium and other organs.
 Historically, hypothermia was the main protective mechanism in cardiac surgery. Currently most cardiac procedures are performed in normothermia or slight hypothermia.
Historically, hypothermia was the main protective mechanism in cardiac surgery. Currently most cardiac procedures are performed in normothermia or slight hypothermia.
The degree of hypothermia can be divided as follows (according to Wong):
| slight |
35 °C – 32 °C |
| moderate |
31 °C – 26 °C |
| deep |
25 °C - 20 °C |
| very deep |
19 °C – 14 °C |
Deep and very deep hypothermia are used in operations where the procedure requires a temporary total circulatory arrest (procedures involving he ascending aorta and arch, some paediatric cardiac procedures). During total circulatory arrest, deep hypothermia is the primary protective mechanism preventing damage to the brain and other organs. The length of arrest must not exceed 40-45 minutes, otherwise brain damage can occur.
Contemporarily, there are some technical solutions to protect the brain, thanks to which the hypothermia need not be that deep and the length of total circulatory arrest time can be prolonged. Example is an antegrade perfusion of the brain (using the carotid arteries) or a retrograde perfusion of the brain (a cannula in the superior vena cava, held in place with a tourniquet) . With these methods arrest time can be prolonged to 60-80 minutes.
Inducing hypothermia and restoring normothermia of the body is an actively controlled process. This is done by change of temperature of the circulating blood with the help of heat exchangers and bed warmers. The body temperature can also be manipulated by placing ice in the operating field, by controlling temperature of infused fluids and of the operating room. As a result of uneven temperature change of the body during hypothermic EC, it is essential to monitor core body temperature (central) and peripheral temperature. Core body temperature is monitored in the distal oesophagus, nasopharynx or urinary bladder while the peripheral temperature is often monitored in the rectum. It is also advantageous to measure the temperature of the blood in the arterial and venous lines of the EC. The patient is considered fully warmed up if core body temperature is 37°C and peripheral temperature is above 35°C.
HEPARIN
Before intraoperative introduction of ECC cannulas, adequate anticoagulation must be achieved to prevent blood clotting in the device, especially in the oxygenator. Unfractionated heparin is used for this exact purpose. It is a mixture of mucopolysaccharides with molecular weight from 3000- 30000 Daltons, whose function is to be a cofactor of antithrombin III (inhibitor of Factor II of coagulation cascade- Thrombin).
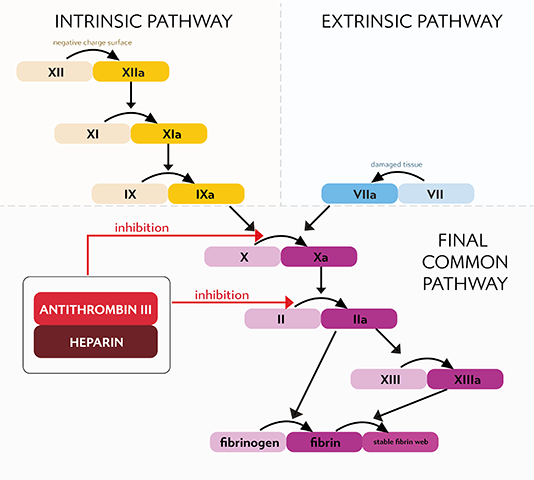
 The level of heparinization during ECC is monitored using ACT test- activated clotting time (the time it takes to form thrombi when blood is in contact with celite surface). Developers of oxygenators often recommend ACT above 480 seconds. To achieve this value, heparin must usually be given at a dose od 2-3 mg.kg-1.
The level of heparinization during ECC is monitored using ACT test- activated clotting time (the time it takes to form thrombi when blood is in contact with celite surface). Developers of oxygenators often recommend ACT above 480 seconds. To achieve this value, heparin must usually be given at a dose od 2-3 mg.kg-1.
The biological half-life of heparin is individual a varies between 1-2 hours. During normothermia, heparin is metabolised faster. During long operations it is required to administer more heparin according to ACT results.
At the end of ECC and after removal of cannulas from the great vessels, protamin is administered to counteract the anticoagulation effects of heparin. Protamin (low molecular protein extracted from the sperm of salmon) is a quick antagonist of heparin, it forms an inert complex with it.
SYSTEMIC INFLAMMATORY RESPONSE
Induction and work of the ECC is, despite continuous improvement, non-physiological challenge for the body. The contact of blood with non-endothelial surface of the tubes and the oxygenator leads to development of systemic inflammatory response. Protein and cellular components of the blood are being activated (Complementary, Kallikrein-kinin, coagulation and fibrinolytics system, activation of thrombocytes, endothelial cells and leucocytes). Apart from it, the body negatively reacts to the effect of heparin and protamine, tissue ischemia and reperfusion, hypothermia, relative hypotension and non-pulsatile blood flow, anaemia, transfusions. The result is SIRS - systemic inflammatory response syndrome. The inflammatory response occurs in a majority of patients after operation without any visible clinical symptoms, it can be however be manifested as post-perfusion syndrome.
Some of the manifestations of post-perfusion syndrome include respiratory failure (ARDS- Adult respiratory distress syndrome), CNS damage (stroke), renal damage (acute renal failure), acute pancreatitis, damage to the GIT (acute ulcer lesions), liver damage (liver failure), coagulation dysfunction (DIC- disseminated intravascular coagulation) or hyperthermia. Significant damage to some other systems can lead to multi-organ damage (MODS- multiple organ dysfunction syndrome) or failure (MOF- multiple organ failure). In modern ECC systems the inner lining of the tubes is coated with various substances to reduce inflammatory response. The mechanism is to decrease the activation of coagulation and other blood humoral systems. The most popular coating is heparin, which also showed promising clinical results.
Components of ECC
VENOUS DRAINAGE
Venous drainage is the transfer of desaturated blood from the patient to the ECC device. Blood is transferred to the machine with the aid of gravity (the height difference between the operation table and the lowly placed venous reservoir) or active suction mechanism (VAVD, further explained).
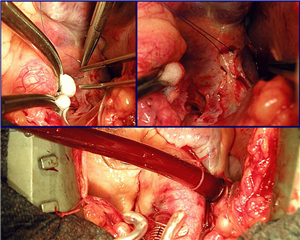
 Technically the venous drainage is achieved by introducing the venous canula into the central venous system. The canula is placed through the right atrium into the superior and inferior vena cava across incisions secured with circular stitches. This type of venous drainage allows the flow of blood around the canula into the right atrium (partial extracorporeal circulation). This type of setup does not allow a bloodless operation field in the right part of the heart. If we place tourniquets around the two caval veins, the amount of blood flowing around the canula is decreased and a setup known as total extracorporeal circulation is created. All the blood coming to the heart is transferred to the ECC. Venous drainage can also be introduced through a peripheral route, mostly into the femoral, and/or into the jugular vein.
Technically the venous drainage is achieved by introducing the venous canula into the central venous system. The canula is placed through the right atrium into the superior and inferior vena cava across incisions secured with circular stitches. This type of venous drainage allows the flow of blood around the canula into the right atrium (partial extracorporeal circulation). This type of setup does not allow a bloodless operation field in the right part of the heart. If we place tourniquets around the two caval veins, the amount of blood flowing around the canula is decreased and a setup known as total extracorporeal circulation is created. All the blood coming to the heart is transferred to the ECC. Venous drainage can also be introduced through a peripheral route, mostly into the femoral, and/or into the jugular vein.
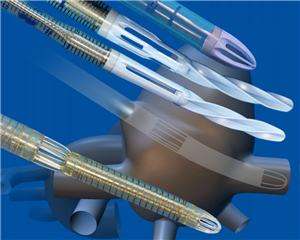 Other setup options include a single two-stage venous canula, which is placed into the auricle of the right atrium- it has a single hole at the end which drains blood from the inferior vena cava and a second proximal hole which drains blood flowing into the atrium from the superior vena cava. Nevertheless, this setup does not allow total extracorporeal circulation.
Other setup options include a single two-stage venous canula, which is placed into the auricle of the right atrium- it has a single hole at the end which drains blood from the inferior vena cava and a second proximal hole which drains blood flowing into the atrium from the superior vena cava. Nevertheless, this setup does not allow total extracorporeal circulation.
Assisted venous return
During the standard set up of the ECC, venous drainage to the reservoir is passive with the help of gravity. To maintain adequate flow of blood it is necessary to use a relatively wide venous cannula. Often however venous drainage can be insufficient. This situation can be solved by adding an active suction module to the venous part of the system.
 The VAVD system (vacuum assisted venous drainage) works on the principle of maintaining controlled negative pressure above the level of blood in the venous reservoir. A specially designed rigid reservoir is connected with a source of controlled negative pressure.
The VAVD system (vacuum assisted venous drainage) works on the principle of maintaining controlled negative pressure above the level of blood in the venous reservoir. A specially designed rigid reservoir is connected with a source of controlled negative pressure.
RESERVOIR
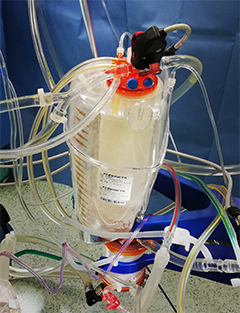 The venous reservoir acts as a storehouse of blood for the entire system and is placed in front of the arterial pump (representing the “atrium”). It is used for gravitational drainage of venous blood from patient's circulation and for balancing fluctuations between the venous inflow and current needs of blood supply to the pump. Bubbles in the reservoir are removed with the help of a filter and drugs can be applied directly into the reservoir, as well. Two types of reservoirs are used – non-rigid and rigid.
The venous reservoir acts as a storehouse of blood for the entire system and is placed in front of the arterial pump (representing the “atrium”). It is used for gravitational drainage of venous blood from patient's circulation and for balancing fluctuations between the venous inflow and current needs of blood supply to the pump. Bubbles in the reservoir are removed with the help of a filter and drugs can be applied directly into the reservoir, as well. Two types of reservoirs are used – non-rigid and rigid.
- Non-rigid venous reservoir - used in the closed system. After filling the device for extracorporeal circulation, air is aspirated from the reservoir and the reservoir is filled solely with blood. When all of the blood is drained from the reservoir, the outflow from the reservoir is blocked by the collapse of a plastic wall (thus preventing air intake). In addition to this, the system must also include another so-called cardiotomy reservoir which collects blood suctioned from the surgical field and heart cavities.
- Rigid reservoir - used in the open system. It is opened to the surrounding atmospheric pressure (unless VAVD is required). It includes a cardiotomy reservoir which is at least partially separated from the venous part. The risk of an open system is air embolization when the reservoir is drained. Therefore, there must always be a minimum amount of blood in the system at all times.
- Cardiotomy reservoir - is a collection place for blood suctioned from the surgical field and so-called vents (suction cannulas introduced into heart cavities for suction of excessive blood filling). Blood from the cardiotomy reservoir is filtered before entering the venous reservoir. This is necessary due to the presence of tissue particles, fat, bone particles, suture material, calcium, fibrin, blood clots, microaggregates or bubbles in the suctioned blood.
PUMP
The pump is the essential part of the ECC device. It replaces the heart in the circulatory system - pumps the desaturated blood from the venous part of the ECC to the oxygenator and arterial part. Requirements of a pump are following:
- sufficient pumping capacity (maintaining the calculated cardiac output)
- technically reliable performance
- gentleness to blood elements.
Two types of pumps are currently used in clinical practice:
 Roller pump
Roller pump
The oldest and most widespread pump. The principle is to move blood in an elastic tube with 2 rotating rollers positioned opposite to each other (180°) that compress the lumen of the tube. The amount of blood flow is determined by the width and length of the tube inserted into the stator and the number of rotations of the motor. It is important to correctly set the pressure of the rollers on the tubes (occlusion), so that sufficient blood flow is maintained and mechanical destruction of blood elements is avoided.
 Centrifugal pump
Centrifugal pump
The principle is the gradual acceleration of blood flowing into the pump using a rotating smooth cone or rotating blades. At the moment of reaching the highest speed on the cone circumference, the blood is drained by centrifugal force into the outflow tube. In case of a technical obstruction in the outflow or inflow there is no danger of a crash - the pump is not able to critically increase pressure. Its performance is reliable and gentle, with a minimal risk of microbubble embolization. Lower levels of hemolysis, activation of blood elements and humoral systems in the blood have been proved. The disadvantage is high price of a single use device.
OXYGENATOR
 An essential component of the ECC that acts as a “lung”. It serves to regulate the exchange of gases between the patient's blood and the supplied gas mixture. The functional capacity of the oxygenator is proportional to the size of the surface on which gas exchange occurs over a concentration gradient.
An essential component of the ECC that acts as a “lung”. It serves to regulate the exchange of gases between the patient's blood and the supplied gas mixture. The functional capacity of the oxygenator is proportional to the size of the surface on which gas exchange occurs over a concentration gradient.
Construction types:
- bubble type- gas exchange occurs during direct contact of blood with O2 (bubbling). It is no longer used (microembolization, traumatization of blood elements, inflammatory response).
- membrane type- blood and gas mixture is separated by a membrane with a large surface area (2 - 5 m2). In the ECC circuit it is placed after the pump because of greater resistance that requires certain blood pressure. There are 2 basic types:
- True membrane oxygenator - the membrane is made of silicone, which is non-porous, gases penetrate only by diffusion. The advantage is good long-term performance, the disadvantage is a large filling volume, a large foreign surface and a high price. The oxygenator is suitable for long-term artificial oxygenation of the patient (ECMO).

- Hollow fibre oxygenator - fibres are made from microporous polypropylene (pore size less than 1 µm). The fibres form a kind of capillary "network" mimicking the pulmonary vascular bed. The pore size does not allow the passage of neither gases nor plasma and blood elements. They are used in routine cardiac surgery. The disadvantage is the reduction of the functional capacity after several hours. The fibres are arranged in a three-dimensional network, with gas flowing intraluminally and blood extraluminally. The amount of CO2 and O2 in the blood can be regulated by adjusting the gas flow rate through the device, or by changing the O2 fraction in the gas mixture. The efficiency of the oxygenator depends on the rate of blood flow through the oxygenator, its viscosity, the external diameter of hollow fibres, the amount of dead space between the fibres, and a number of other design factors.
HEAT EXCHANGER

 The heat exchanger provides cooling and heating of the circulating blood in the ECC. It is part of every oxygenator and is located in its venous section in front of the oxygenation unit. On one side of its surface is blood and on the other side circulating water. The compartments are separated by a thermally conductive material (anodized aluminum, stainless steel or plastics).
The heat exchanger provides cooling and heating of the circulating blood in the ECC. It is part of every oxygenator and is located in its venous section in front of the oxygenation unit. On one side of its surface is blood and on the other side circulating water. The compartments are separated by a thermally conductive material (anodized aluminum, stainless steel or plastics).
An external heating unit (“heater- cooler unit”) is required for the function of the exchanger. It is a source of a water of precisely selected temperature. This is led through the tubes to the heat exchanger. Here it flows through a system of spiral or thin tubes - the temperature of the circulating blood is regulated through their walls.
 ARTERIAL CANULLA
ARTERIAL CANULLA
The arterial cannula is used to return oxygenated blood from the ECC back to the patient's arterial bloodstream. It must meet demanding design requirements due to its location in the high-pressure part of the ECC and due to connection to the patient's arterial bloodstream.
The most common site of arterial cannula insertion is the distal part of the ascending aorta before the brachiocephalic trunk. Under special circumstances, this cannula can be inserted into the femoral or axillary artery. The cannulas are specially adapted for these setups.
ADDITIONAL EQUIPMENT
- Vent
It is a suction cannula that is inserted into a heart chamber during extracorporeal circulation. The aim is to prevent excessive distension (especially of the left ventricle), to ensure a bloodless and clear surgical field and to facilitate de-airing of the heart at the end of the operation.
- Coronary suction
An additional suction that allows the surgeon to drain blood from the surgical field directly into the ECC - the cardiotomy reservoir. It is the largest source of haemolysis, corpuscular and gas embolization.
- Filters
The most important is arterial filter, which is inserted into the arterial line beyond the oxygenator. Structurally, it is a network of polyester fibres with a pore diameter of 20 - 40 µm. It is able to eliminate both corpuscular micro-embolisms and gaseous embolisms (“bubble trap”), the most dangerous source of this being coronary suction.
- Haemoconcentrator
Device designed for immediate removal of excess crystalloids from perfusate and thereby increasing the haematocrit of circulating blood. It separates the cellular and protein components of blood from water and low molecular weight substances in plasma on the principle of ultrafiltration through a semipermeable membrane. Indications for its use include a decrease in haematocrit during extracorporeal circulation (below 0.20), preoperative hyperhydration due to heart failure, prolonged extracorporeal circulation, and known decrease in renal function. It is usually placed behind the arterial filter.
Management of ECC
PREPARATION BY THE PERFUSIONIST
The perfusionist - the person who operates the heart-lung machine and cardiopulmonary bypass – must be acquainted with the type of operation and the patient's documentation. On this basis, he chooses the appropriate system type and cannula. He calculates patient's body surface area, required systemic flow rate during the ECC and filling volume of the entire system to avoid excessive hemodilution. A record of the whole perfusion is filled in, as well. The system is filled with replacement solutions and de-aired. Afterwards, the system is ready to be connected with patient's vascular system.
PREPARATION OF THE PATIENT
After sternotomy, heparin is administered to the patient. The surgeon prepares the target structures for the insertion of the cannulae (most often the ascending aorta and the right atrial appendage). Circular stitches with tourniquets are placed in there. Once effective heparinization (according to ACT) is achieved, the surgeon inserts a cannula through a point incision in the aorta. Subsequently it is connected to the EC arterial line in an air-free manner. The procedure is similar for the venous canula in the right atrium.
INITIATION AND MANAGEMENT OF EXTRACORPOREAL CIRCULATION
The ECC is initiated by the command of the operating surgeon. Clear communication between the surgeon and the perfusionist is important. After the arterial line is released, the perfusionist slowly increases the pump speed and at the same time slowly releases the flow through the venous line so that blood from the patient begins to flow to the EC system. The pump speed is increased so that it‘s performance gradually reaches the required calculated blood flow. The perfusionist opens the gas flow to the oxygenator and eventually begins to cool the patient to the required temperature. Subsequently, it is possible to proceed with the surgery itself on a stopped heart.
Continuous monitoring during ECC
During ECC, blood flow through the pump, pressure in the ECC system, blood pressure, patient's temperature in the oesophagus and rectum (or bladder), arterial blood temperature, venous blood temperature, water temperature, O2 saturation in the venous line, pO2 are monitored. The level of heparinization must also be checked at regular intervals.
TERMINATION OF EXTRACORPOREAL CIRCULATION
A while before the expected completion of the heart procedure, the surgeon instructs the perfusionist to start rewarming of the patient. The ECC can only be stopped when normothermia is re-established. After the recovery of heart activity and after the phase of alleviated circulation (phase of myocardial reperfusion), the termination of ECC can be considered. The perfusionist gradually slows down the return of venous blood from the patient until the venous line is completely closed. At the same time, the pump speed is slowed down to gradually return most of the blood from the ECC system to the vascular bed. Arterial pressure and heart filling are monitored, as well. When the blood pressure is normalized and the heart is optimally filled, the ECC can be completely stopped.
At the end of the procedure, practically the entire blood volume of the ECC system is gradually reinfused back to the patient's vascular bed and the effect of heparin is antagonised by intravenous administration of protamine.
Myocardial protection
DEFINITION
Most of the procedures in cardiac surgery on ECC are feasible only during temporary withdrawal of the heart from the circulation. After the induction of cardiopulmonary bypass, a vascular clamp is placed across the ascending aorta (so called aortic cross-clamp). This completely interrupts the flow of oxygenated blood to the coronary arteries, the period of global myocardial ischaemia begins.
There is a number of pathophysiological events that occur during normothermic ischemia of the cardiomyocytes - from reversible systolic-diastolic dysfunction (myocardial stunning) to myocyte necrosis (20 minutes to several hours). Due to the risk of fatal perioperative myocardial infarction, many procedures were developed to maintain myocardial viability during the period of global ischaemia and to fully restore its functionality in the phase of reperfusion.
Most of these procedures have in common to minimize myocardial metabolic demands during ischaemia. Decreasing the metabolic needs of myocytes allows the heart to survive the phase of global myocardial ischemia without clinically significant damage.
 HYPOTHERMIA
HYPOTHERMIA
Hypothermia reduces myocyte energy requirements and thus enables the myocardium to tolerate the period of global myocardial ischemia, however it is standalone not sufficient to protect the cardiac muscle for the time normally required to perform cardiac surgery. The protective effect of hypothermia is used in combination with cardioplegic cardiac arrest. Cooling of the heart muscle (usually 8 - 10 ° C) is achieved by three factors:
- whole-body mild hypothermia
- local cooling of the heart by ice
- perfusion of the coronary bed with a cardioplegic solution at 4 ° C
Continuous re-warming of the heart (from the body core and non-coronary flow) is prevented by repeated administration of cold cardioplegic solution, and eventually local ice application.
CARDIOPLEGIA
The best way to protect the cardiomyocytes and to preserve their intracellular energy sources is to immediately stop myocardial electromechanical activity as soon as the coronary flow is interrupted after the aortic cross-clamp. This can be achieved by administration of cardioplegic solution into the coronary bed. The principle of its function is to induce asystole in the diastolic phase of heart cycle with the help of high potassium concentration in the administered solution (15-30 mmol / l).
Hyperkalemic cardiac arrest in the diastole works by changing the resting membrane potential of cardiomyocytes from the original -85 mV to approximately -60 mV. This is achieved due to a decrease in the transmembrane gradient of potassium concentration. Above this value, the rapid sodium channels (required to trigger the action potential,and hence myocardial excitement and contraction) can no longer be activated. In addition to this, changes in concentration of other ions facilitate the cardiac arrest in various degrees, depending on the type of solution selected.
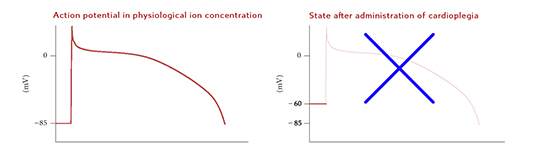
According to ion composition in the solution, we distinguish intracellular solutions having a low sodium and calcium concentration and extracellular solutions with high concentration of these ions.
| Type of solution |
Na+ |
K+ |
Mg2+ |
Ca2+ |
pH |
| St Thomas II (extracellular) |
120 |
16 |
16 |
1.2 |
7.8 |
| Custodiol (intracellular) |
15 |
9 |
4 |
0.015 |
7.2 |
| Blood cardioplegia 4:1 (initial dose) |
140 |
20 |
13 |
- |
7.2 |
| Blood cardioplegia 4:1 (repeated dose) |
140 |
10 |
9 |
- |
7.4 |
Cardioplegia can be either crystalline (always cold, administered as an aqueous ion solution) or blood (concentrated cardioplegia solution mixed with oxygenated blood). Blood cardioplegia provides better protection of the ischemic myocardium (erythrocyte oxygen supply, blood buffering ability, oncotic activity of plasma proteins). According to the temperature of blood cardioplegia we distinguish cold (4 - 10 ° C), warm (normothermic) and tepid (35 ° C).
 Cardioplegia application options
Cardioplegia application options
Cardioplegic solution is administered antegrade (into the aortic root, directly to the coronary orifices, into the vein bypass) or retrograde via the coronary sinus (better distribution in case of significant diffuse coronary atherosclerosis), or both. The administration of the solution should be repeated every 20-40 minutes, except for specially prepared solutions where a single initial dose covering a longer period of time (90-120 minutes) is sufficient – e.g. Del Nido, Custodiol, etc..
BETAPLEGIA
A state simmilar to cardioplegic arrest can be induced pharmacologically by the administration of a short-acting beta-blocker esmolol into the ascending aorta. Inactive myocardium is perfused with normothermic blood throughout the cardiac arrest.
OTHER TECHNIQUES
Depending on workplace traditions, some other methods of myocardial protection can be used during the period of global myocardial ischemia:
Intermittent aortic cross-clamping
It is used in coronary surgery. In extracorporeal circulation with mild hypothermia (33 ° C), the surgeon induces ventricular fibrillation with a fibrillator device and then places the cross clamp on the aorta. The heart fibrillates during peripheral coronary anastomosis. Upon completion of the peripheral anastomosis, the surgeon releases the aortic cross clamp and defibrillates the heart. During the reperfusion period, he sews the proximal anastomosis on the ascending aorta and the whole procedure may be repeated. Although ventricular fibrillation is energetically disadvantageous even under hypothermia, the clinical experience with this procedure is good.
Total cardiac arrest in deep hypothermia
After introduction of extracorporeal circulation, the patient's temperature is gradually decreased to 15 - 20 ° C. Cooling can be facilitated by lining the body with ice bags. The cardiac procedure is performed in total cardiac arrest with an expected safe period of 45 min. This technique is used in paediatric cardiac surgery.
Resources:
- Lonský V. Mimotělní oběh v klinické praxi. Praha: Grada Publishing, 2004. ISBN 80-247-0653-9.
- Wagner R. Kardioanestezie a perioperační péče v kardiochirurgii. Praha: Grada Publishing, 2009. ISBN 978-80-247-1920-7.
- Scarola C. Cardiopulmonary Bypass and Biocompatibility: A Review. Journal of Biomaterials Applications Reviews. p 42-58. Vol 4. 2010
- Angelo FM. "Dr. John H. Gibbon, Jr. and Jefferson's Heart-Lung Machine: Commemoration of the World's First Successful Bypass Surgery". Posters: Jefferson History. 2015. http://jdc.jefferson.edu/jeffhistoryposters/1
- Habertheuer A, Kocher A, Laufer G, Andreas M, Szeto WY, Petzelbauer P, et al. Cardioprotection: a review of current practice in global ischemia and future translational perspective. BioMed research international. 2014;2014:325725.
- Dominik J. Kardiochirurgie. Praha: Grada Publishing, 1998. ISBN: 80-7169-669-2.
- Dominik J.: Kardiochirurgie. In.: Pafko P. et al.: Základy speciální chirurgie, Galén, 2008, s.173-195. ISBN 978-80-7262-402-7
We hereby thank Martin Volt, the leader of perfusionists team at Cardiac Surgery Department in University Hospital Hradec Kralove, for the text control, practical insight and photographic illustrations.
