Aortic Valve Disease

Aortic valve is a semilunar valve placed at the end of left ventricualr outflow tract. It separates left ventricle from aorta. It prevents retrograde blood flow from the aorta to the left ventricle in diastole, and at the same time enables unresisted blood outflow from the heart to the systemic circulation. Correct function of aortic valve is a requirement for an error-free performance of the heart. The diseases of aortic valve can be divided from hemodynamic point of view into stenosis, regurgitation and combined disease. Combined disease carries the signs of both previous options in various range, depending on which component is dominant.
Etiology and Pathogenesis
Aortic valve stenosis is a disease caused by disorder of aortic valve cusps opening in systole. A resistance arises in blood flow from the left ventricle to the aorta. An afterload of left ventricle increases, which results in pressure-overload. Compensatory mechanism is a concentric hypertrophy of left ventricle, that is, however, connected with diastolic dysfunction. Because of myocardial hypertrophy and increased left ventricular diastolic pressure, the coronary perfusion is affected. This can lead to systolic dysfunction of left ventricle. Late stage of the disease is characterised by low ejection fraction of the left ventricle, left heart failure and pulmonary hypertension.
Aortic valve stenosis is the most common valve disease of adults in western countries. Its incidence rises with age and is more frequent in males. In the population over 65 years the prevalence is 2%. It has basically two causes:
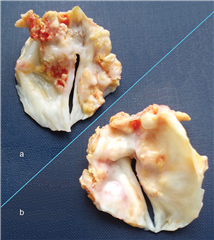
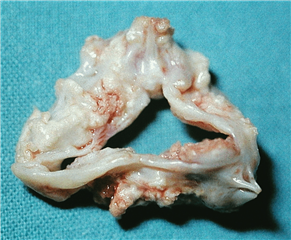
- congenital heart valve disease in terms of bicuspid or unicuspid aortic valve. It may manifest in the first hours after surgery when critical. Or it may stay asymptomatic for years and becomes evident firstly after 40 years of age due to its higher susceptibility to degeneration. Besides this valvular form, there are also subvalvular and supravalvular variants of congenital aortic valve stenosis.
- degenerative aortic valve calcification of the elderly. It is a calcification of aortic valve cusps that imitates atherosclerosis. In late stage the cusps are rigid and calcifications grow into the valve anulus, aortic root, even into the anterior leaflet of the mitral valve. Adhesions of commisures are not present. It usually arises after 60 years of age.

- rheumatic aortic valve stenosis was the most frequent heart valve disease in the past together with rheumatic mitral valve disease. Thanks to the eradication of rheumatic fever in developed countries and progress in antibiotic therapy, it is currently rare. It is characterised by rigid stenotic to stenoinsufficient valve ostium with adhesions in commisures.
- other rare causes: rheumatoid heart disease, familiar hypercholesterolemia, lupus erythematosus, ochronosis.
Aortic valve regurgitation is characterised by a diastolic reflux of the blood from the aorta to the left ventricle. It is caused by insufficient coaptation of aortic valve cusps. It leads to volume-overload of the left ventricle toghether with high preload. When the regurgitation develops slowly, the ventricle is able to adapt to this condition for long time and manifests as an excentric hypertrophy. On the basis of Frank-Starling law the stroke volume increases in dilated ventricle and the patient is asymptomatic in the long-term. By further development of the valve disease, the reserves of compensatory mechanisms become exhausted, which leads to decrease in left ventricular ejection fraction and to left heart failure. Clinical symptoms arise not until this late stage. This may be often irreversible, therefore the aortic valve regurgitation is consideres „insidious“.
When the aortic valve regurgitation develops in acute settings, compensatory mechanisms apply only in a limited extent. Due to low adaptability of the left ventricle, volume-overload arises together with pressure-overload. This affects the left atrium and pulmonary circulation, as well. Acute heart failure with pulmonary edema results.
Aortic root consists of 4 functional components: aortic valve anulus, cusps of aortic valve, sinuses of Valsalva, sinotubular junction (STJ). The regurgitation originates from dilation of anulus or STJ, from redundancy (prolapse) or deficiency (retraction) of cusp tissue. The sinuses of Valsalva do not play a role in aortic valve competency, their function is to minimise mechanical stress on cusps during heart cycle.
Chronic aortic valve regurgitation can be caused by:
- congenital heart valve disease - bicuspid or unicuspid valve, similar to stenosis. The lower tissue quality leads to dilation of anulus and cusp prolapse.
- sclerotic and calcificative degeneration - retraction of the cusps with subsequent insufficient coaptation.
- myxoid degeneration - prolapse of redundant myxoid-degenerated cusps.
- post-rheumatic disease - on the basis of cusp retraction.
- dilation of the aortic root (so called anuloaortic ectasia) - leads to central regurgitation, mostly in genetic connective tissue disorders (Marfan or Ehlers-Danlos syndrome).
- status post infective endocarditis - after healing the infection, defects in the cusps can arise and lead to deformation and malfunction of the valve.
- infective and non-infective aortitis - syphilitic, Reiter syndrome, rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus
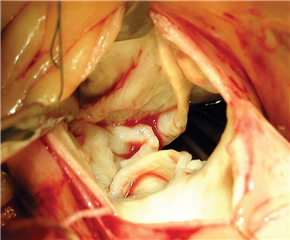
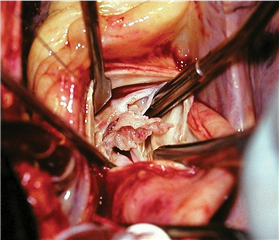
Acute aortic valve regurgitation has following causes:
- infective endoarditis - perforation and destruction of the cusps due to infection process
- type A aortic dissection - false canal in the aortic wall can tear a commisure of aortic valve that subsequently prolapses into the left ventricle together with the cusps
- trauma - rare cause, aortic valve is teared by deceleration forces
Diagnosis
SYMPTOMS
Aortic valve stenosis. Fully developed disease is characterised by pathognostic triad:
- dyspnoea on exertion
- chest pain
- syncope / presyncope
In late stage the signs of left (and right) heart failure can be present.
Aortic valve regurgitation. Chronic disease stays asymptomatic for long. In decompensated stage the symptoms of post-capillary pulmonary hypertension are present - dyspnoea, ortopnoea, paroxysmal nocturnal dyspnoea, pulmonary edema. Chest pain is present, as well. Syncopes are rare in comparison with aortic valve stenosis.
Acute aortic valve regurgitation has a dramatic course, manifesting itself by an acute heart failure, up to cardiogenic shock.
PHYSICAL EXAMINATION
Stenosis:
- ejection systolic murmor over the aortic valve with propagation to the carotids
- peripheral pulse with low amplitude and sustained upstroke (pulsus parvus et tardus)
- signs of congestive heart failure and pulmonary edema in late stage
Regurgitation:
- diastolic „decrescendo“ murmur over upper third of breastbone, with a peak in Erb’s point, propagating to the apex of the heart
- great systolic-diastolic amplitude of peripheral pulse (Corrigan pulse, pulsus altus et celer)
- systolic nodding of patient’s head while sitting, synchone with heartbeat (de Musset‘s sign)
- pulsations of uvula (Müller’s sign)
- capillary pulsation on lips and fingers (Quincke’s sign)
ECG
Stenosis manifests as hypertrophy and overload of the left ventricle, deviation of heart axis to the left. Ventricular dysrhytmias can be present at late stage. Regurgitation is of similar image.
X-RAY
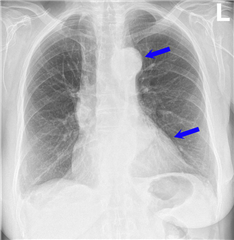 Cardiomegaly is typical for both options of aortic valve diseas. Calcifications of the valve and aorta are sometimes to be seen. Aortic knob can be prominent in dilation of the aorta.
Cardiomegaly is typical for both options of aortic valve diseas. Calcifications of the valve and aorta are sometimes to be seen. Aortic knob can be prominent in dilation of the aorta.
ECHOCARDIOGRAPHY
Gold standard to prove presence and significance of aortic valve disease. Transthoracic ECHO is usually sufficient. If the finding is unclear or a valve-sparing procedure is considered, transoesophageal ECHO is required.
Significant aortic valve stenosis carries the following ECHO signs:
- significant pathomorphology of the valve
- mean transvalvular pressure gradient ΔPm ≥ 40 mmHg
- transvalvular peak velocity Vmax ≥ 4 m . s-1
- aortic valve area AVA ≤ 1.0 cm2
- aortic valve area indexed to the body surface area AVAi ≤ 0.6 cm2 . m-2
Significant aortic valve regurgitation carries the following ECHO signs:
- cusp coaptation defect
- regurgitation jet width takes >65% of the left ventricular outflow tract
- vena contracta (jet width in the narrowest place) over 6mm
- regurgitant fraction >50%
- effective regurgitant orifice area >0.30 cm2
- holodiastolic flow reversal in dsecending aorta

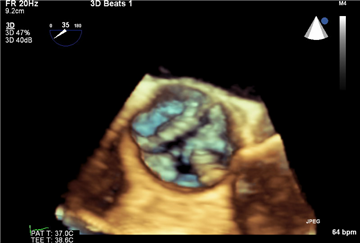

In unclear cases, especially in asymptomatic aortic stenosis, stress-dobutamin echocardiography may be helpful. Besides the ECHO changes, the typical triad of symptoms or abnormal blood pressure drop can occur. In these cases the disease is considered symptomatic.
HEART CATHETERIZATION
It plays only a supplementary role in stenosis, as well as in regurgitation. It may be used to visualize coronary arterial bed due to the fact that coronary artery disease is very common in patients with degenerative aortic valve disease. The direct measurement of transvalvular pressure gradient or regurgitant fraction is possible in case of uncertain ECHO finding.
COMPUTED TOMOGRAPHY
It is not suitable for primary diagnostics. It may be used to quantify the amount of aortic valve calcification (i.e. Calcium score) that is helpful in some borderline indications for surgery. It is of particular importance in pre-procedural planning of transcatheter aortic valve implantation (TAVI).
MAGNETIC RESONANCE IMAGING
It may be used in case of insufficient ECHO quality or in discrepant findings. Particularly suitable for evaluation of the severity of aortic valve regurgitation. It can perfectly describe heart chamber volumes, function, abnormities of ascending aorta or myocardium (e.g. fibrosis). The disadvantage is high time and financial demand, as well as availability of the method.
Therapy
Pharmacological treatment of aortic valve disease does not bring essentially any significant benefit. The exceptions are some speficic illnesses, where it has an irreplaceable function: infective endocarditis (antibiotics), congenital connective tissue disorders and aortic dissection (beta-blockers and antihypertensives), autoimmune diseases (imunosupression). The only causal treatment of aortic valve disease is surgery, either valve replacement, plasty or transcatheter implantation of bioprothesis (TAVI).
INDICATIONS FOR SURGERY
Aortic valve stenosis is indicated for surgery in symptomatic patients with proven severe stenosis. When the patient is asymptomatic, the severe valve disease is indicated for surgery after positive stress testing, after proving low ejection fraction of the left ventricle (<50%), or in case of concomitant cardiac surgery procedure (severe disease of other valve or aorta, coronary artery bypass grafting). In concomitant procedures, a moderate aortic valve stenosis may be operated on, too. An indication problem arises in so called „low-flow, low-gradient“ aortic valve stenosis - aortic valve area is in the range of severe disease, but the transvalvular gradient is below the significant value. While considering the surgery, the stress echocardiography or CT evaluation of aortic valve calcium burden can be helpful.
Aortic valve regurgitation is indicated for surgery in severe valve disease. The intervention is always justified in symptomatic patients. In asymptomatic ones the valve can be operated on in case of low ejection fraction of the left ventricle or together with concomitant cardiac surgery procedure. Surgery should be considered in asymptomatic patients with preserved ejection fraction when severe dilation of the left ventricle is present (end-systolic diameter >50mm, end-diastolic diameter >70mm). ¨
A special indication for surgery is infective endocarditis of aortic valve. Effective antibiotic treatment is a keystone of patient management. The surgery is to proceed in following circumstances:
- congestive heart failure (because of underlying aortic valve regurgitation, obstruction or fistula)
- acute dysfunction of the valve
- globally uncontrolled infection (persistent sepsis despite adequate antimicrobial treatment)
- locally uncontrolled infection (formation of an abscess, fistula or pseudoaneurysm)
- massive valve vegetations, especially already after embolisation events
AORTIC VALVE REPLACEMENT
Aortic valve replacement is the most common cardiac valve procedure in adults in modern society. The principle is to remove native diseased valve, to handle local damage (decalcification of the anulus, drainage of abscesses etc.), and to implant a new valve. Nowadays mechanical and biological valves are at disposal. The following variants of biological valves are being used:
- biological stented prosthesis (made of bovine pericardium or porcine aortic valve cusps)
- biological stentless prosthesis
- biological sutureless prosthesis
- homograft (cryopreserved aortic root from a cadaverous human donor)
- conduit - composite prosthesis of aortic valve and ascending aorta (i.e. Bentall procedure)
- autograft (pulmonary root of the patient himself, ie. Ross procedure)

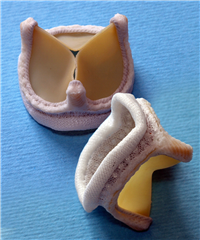 Mechanical and biological stented prostheses are currently the two most widespread options. Mechanical prosthesis is historically older and most preferred until recently. It has an advantage of an excelent long-term durability and good hemodynamic attributes. Nevertheless, patient is limited by lifelong anticoagulation treatment (due to the risk of thrombosis and embolization). In inadequate compliance is the patient exposed to the thrombembolic, as well as bleeding complications. Stented bioprosthesis is easy to implant and patient is not limited by lifelong anticoagulation treatment. On the other hand, the valve has worse hemodynamic specifications (smaller efective orifice area in comparable valve diameter) and it shows a tendency to degenerate, particularly in younger patients or in patients with renal failure or hyperparathyreoidism. Patients are at risk of demanding re-do surgery (re-replacement of the valve). Recently the longevity of the bioprostheses rises thanks to anticalcification adjustment. Recommended age limit to implant bioprosthesis is currently 60-65 years (below this limit the mechanical prostheses are implanted).
Mechanical and biological stented prostheses are currently the two most widespread options. Mechanical prosthesis is historically older and most preferred until recently. It has an advantage of an excelent long-term durability and good hemodynamic attributes. Nevertheless, patient is limited by lifelong anticoagulation treatment (due to the risk of thrombosis and embolization). In inadequate compliance is the patient exposed to the thrombembolic, as well as bleeding complications. Stented bioprosthesis is easy to implant and patient is not limited by lifelong anticoagulation treatment. On the other hand, the valve has worse hemodynamic specifications (smaller efective orifice area in comparable valve diameter) and it shows a tendency to degenerate, particularly in younger patients or in patients with renal failure or hyperparathyreoidism. Patients are at risk of demanding re-do surgery (re-replacement of the valve). Recently the longevity of the bioprostheses rises thanks to anticalcification adjustment. Recommended age limit to implant bioprosthesis is currently 60-65 years (below this limit the mechanical prostheses are implanted).
 Biological stentless prosthesis is an useful option for patients, to whom we want to implant a bioprosthesis, however, they have unsuitable anatomy - very narrow valve anulus. Small stented bioprosthesis would not practically help the patient and would keep high transvalvular gradient. This situation is called „patient-prosthesis mismatch“ and it can be counted as efective orifice area of the prosthesis divided by patient’s body surface area. The mismatch is present in value <0.85 cm2/m2, severe mismatch is below 0.6 cm2/m2 (of note is the analogy with ECHO criteria of aortic valve stenosis severity). In this case it is very probable that patient will not benefit from the surgery and the prosthesis will soon degenerate. The stentless prosthesis offers better hemodynamic attributes, even in smaller anulus, thanks to omiting the sewing ring. Other option for the patient would be implantation of mechanical prosthesis or some of the aortic root enlargement procedures. The latter is however technically demanding and is performed rarely nowadays.
Biological stentless prosthesis is an useful option for patients, to whom we want to implant a bioprosthesis, however, they have unsuitable anatomy - very narrow valve anulus. Small stented bioprosthesis would not practically help the patient and would keep high transvalvular gradient. This situation is called „patient-prosthesis mismatch“ and it can be counted as efective orifice area of the prosthesis divided by patient’s body surface area. The mismatch is present in value <0.85 cm2/m2, severe mismatch is below 0.6 cm2/m2 (of note is the analogy with ECHO criteria of aortic valve stenosis severity). In this case it is very probable that patient will not benefit from the surgery and the prosthesis will soon degenerate. The stentless prosthesis offers better hemodynamic attributes, even in smaller anulus, thanks to omiting the sewing ring. Other option for the patient would be implantation of mechanical prosthesis or some of the aortic root enlargement procedures. The latter is however technically demanding and is performed rarely nowadays.
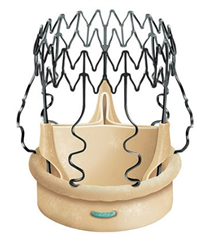 Biological sutureless prosthesis is a novel technological variant of bioprosthesis. Without the need of stitches, the prosthesis is placed into the aortic valve position where it is held by self-expandable (nitinol stent) or balloon-expandable mechanism. Sutureless prostheses offer comparable hemodynamic attributes to stentless prostheses, nevertheless, the implantation is less time- and technically demanding than in any other option. The disadvantage is higher price, a risk of paravalvular leakage and of damage to cardiac conduction system.
Biological sutureless prosthesis is a novel technological variant of bioprosthesis. Without the need of stitches, the prosthesis is placed into the aortic valve position where it is held by self-expandable (nitinol stent) or balloon-expandable mechanism. Sutureless prostheses offer comparable hemodynamic attributes to stentless prostheses, nevertheless, the implantation is less time- and technically demanding than in any other option. The disadvantage is higher price, a risk of paravalvular leakage and of damage to cardiac conduction system.
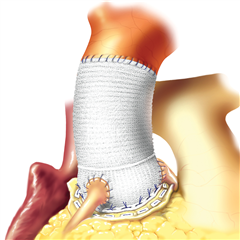
 Aortic valve replacement with homograft is a complex procedure, it is a replacement of the whole aortic root, not only the valve itself. Apart from sewing the graft into the aortic anulus, the reimplantation of coronaries and distal graft-to-aorta anastomosis are required. The longevity of homografts is comparable with standard bioprostheses, though they have better hemodynamic attributes and better resistance to infection - they are particularly recommended in condition of infective endocarditis.
Aortic valve replacement with homograft is a complex procedure, it is a replacement of the whole aortic root, not only the valve itself. Apart from sewing the graft into the aortic anulus, the reimplantation of coronaries and distal graft-to-aorta anastomosis are required. The longevity of homografts is comparable with standard bioprostheses, though they have better hemodynamic attributes and better resistance to infection - they are particularly recommended in condition of infective endocarditis.
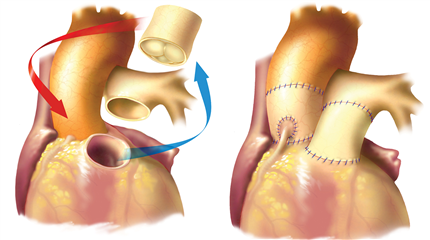
 Bentall procedure is a replacement of aortic valve and diseased ascending aorta with a composite prosthesis - conduit. Mechanical, as well as biological, option is available. It is being used in anuloartic ectasia, or in aortic valve stenosis with aneurysmatic dilation of aortic root and ascending aorta. Similarly as in homograft, it is a complex procedure - apart from sewing the prosthesis into the aortic anulus, reimplantation of coronaries and distal anastomosis of graft to aorta are required.
Bentall procedure is a replacement of aortic valve and diseased ascending aorta with a composite prosthesis - conduit. Mechanical, as well as biological, option is available. It is being used in anuloartic ectasia, or in aortic valve stenosis with aneurysmatic dilation of aortic root and ascending aorta. Similarly as in homograft, it is a complex procedure - apart from sewing the prosthesis into the aortic anulus, reimplantation of coronaries and distal anastomosis of graft to aorta are required.
 Ross procedure is a complex procedure, its principle is to implant patient’s own pulmonary root into the aortic position. Pulmonary homograft is implanted into patient’s pulmonary position. This option has the best hemodynamic attributes, no thrombogenicity and excellent longevity (actually patient’s own living tissue is used). It offers high quality of life, long-term survival of patients is comparable with healthy population. On the other hand, it is time and technically demanding procedure and it rises some controversies in cardiosurgical community, because it makes two-valve disease out of one-valve disease. From the long-term point of view, there is some risk of re-do surgery due to graft dilation or other technical deterioration (approximately 20% in 20 years postoperatively). It is indicated in young active patients below 50 years of age, rarely older. It may be considered in patients with contraindication to warfarin and in young women desiring for pregnancy.
Ross procedure is a complex procedure, its principle is to implant patient’s own pulmonary root into the aortic position. Pulmonary homograft is implanted into patient’s pulmonary position. This option has the best hemodynamic attributes, no thrombogenicity and excellent longevity (actually patient’s own living tissue is used). It offers high quality of life, long-term survival of patients is comparable with healthy population. On the other hand, it is time and technically demanding procedure and it rises some controversies in cardiosurgical community, because it makes two-valve disease out of one-valve disease. From the long-term point of view, there is some risk of re-do surgery due to graft dilation or other technical deterioration (approximately 20% in 20 years postoperatively). It is indicated in young active patients below 50 years of age, rarely older. It may be considered in patients with contraindication to warfarin and in young women desiring for pregnancy.
Valve type choice does not depend on the arbitrary age limit only, but other factors have to be also considered: biological age of the patient, comorbidities, anatomical conditions, the need for anticoagulations, lifestyle. A dialogue with patient plays a key role in this. Advantages and disadvantages of each possible option are explained to the patient and the final decision is up to him.
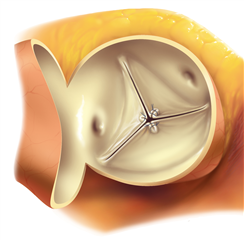
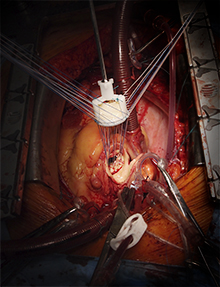 Because of the wide range of the topic, only the surgical principles of standard aortic valve prosthesis implantation will be further explained (mechanical and biological stented prostheses; they are practically the same, except of some little technical details). The surgical approach to the valve is median sternotomy or some of the minimally invasive approaches (upper hemisternotomy, right anterior thoracotomy). The procedure is carried out using extracorporeal circulation. After cross-clamping the ascending aorta the heart is stopped by cardioplegic solution, ascending aorta is opened above the right coronary artery and the diseased valve is excised. Only a narrow edge of anulus is left in situ for comfortable placement of the stitches. Thorough decalcification of the anulus is important, consequently avoiding damage to conduction system, perforation or embolization of calcifications. Optimal valve size is determined using special measurement tools placed into the aortic anulus. Pledgeted matress sutures are anchored in the anulus, their orientation depends on the desire to implant the valve intra- or supra-anularly. The stitches are then passed through the sewing ring of the prosthesis „par distance“. After all the stitches are passed through the aortic anulus of the patient, as well as through the sewing ring of the prosthesis, the prosthesis is placed into the aortic anulus in a parachute manner. The stitches are tied and cut. After checking valve function the heart chambers are de-aired and aortotomy is closed.
Because of the wide range of the topic, only the surgical principles of standard aortic valve prosthesis implantation will be further explained (mechanical and biological stented prostheses; they are practically the same, except of some little technical details). The surgical approach to the valve is median sternotomy or some of the minimally invasive approaches (upper hemisternotomy, right anterior thoracotomy). The procedure is carried out using extracorporeal circulation. After cross-clamping the ascending aorta the heart is stopped by cardioplegic solution, ascending aorta is opened above the right coronary artery and the diseased valve is excised. Only a narrow edge of anulus is left in situ for comfortable placement of the stitches. Thorough decalcification of the anulus is important, consequently avoiding damage to conduction system, perforation or embolization of calcifications. Optimal valve size is determined using special measurement tools placed into the aortic anulus. Pledgeted matress sutures are anchored in the anulus, their orientation depends on the desire to implant the valve intra- or supra-anularly. The stitches are then passed through the sewing ring of the prosthesis „par distance“. After all the stitches are passed through the aortic anulus of the patient, as well as through the sewing ring of the prosthesis, the prosthesis is placed into the aortic anulus in a parachute manner. The stitches are tied and cut. After checking valve function the heart chambers are de-aired and aortotomy is closed.
AORTIC VALVE PLASTY
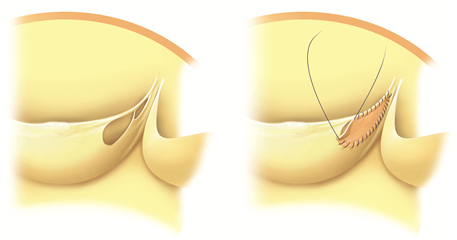
In some patients with aortic valve regurgitation there is no need to replace the valve. Using special surgical techniques it is possible to reestablish reliable cusp coaptation and to preclude retrograde blood flow in diastole. In case of dilated aortic anulus it is possible to reduce its diameter and stabilize it with the help of external ring or special suture anuloplasty. The treatment in dilation of sinotubular junction (often together with dilation of ascending aorta) is the supracoronary ascending aortic replacement by a tubular prosthesis of such size, that keeps adequate cusp coaptation. In cusp prolapse, central cusp plication may be carried out (shortening of the free margin of the cusp by a centrally placed stitch). Other procedures include: oversewing the cusp fenestration by a stitch or pericardial patch, suture of incompletely fused bicuspid valve cusp, removal of redundant tissue and calcifications („shaving“). In aortic valve regurge caused by cusp retraction is the prognosis of plasty uncertain and is not recommended.

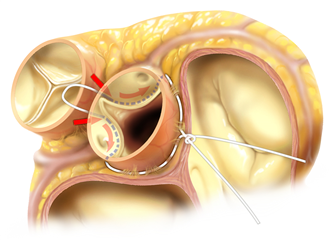
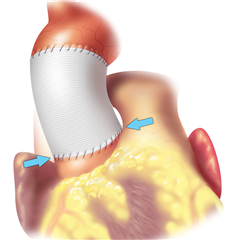
The radical Bentall procedure used to be the gold standard in case of anuloaortic ectasia (dilation of the whole aortic root). Nowadays when possible, aortic valve sparing procedures are being recommended (native cusps are spared). In „neo-aorta“ with physiological diameters, reaching adequate cusp coaptation and removal of regurgitation are anticipated. Yacoub procedure (aortic root remodelling) and David procedure (aortic root reimplantation) belong here. The principle of Yacoub procedure is to replace ascending aorta and sinuses of Valsalva with a prosthesis, and eventually stabilize the anulus and repair the cusps. David procedure consists in pulling of the aortic valve into the tubular prosthesis, that consequently replaces the ascending aorta. By this means the stabilization of anulus, of sinuses of Valsalva and of sinutubular junction is reached. The procedure can be extended by repair of the cusps. These procedures have excellent long-term outcomes and offer good quality of life without the need of anticoagulation. A low risk of re-do surgery remains because of plasty failure.

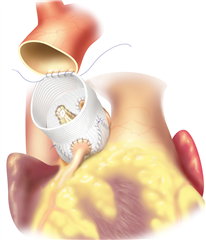
TRANSCATHETER AORTIC VALVE IMPLANTATION (TAVI)
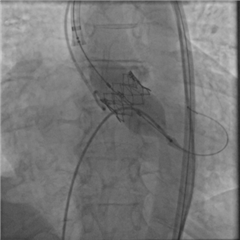
 TAVI represents a distinct approach in the treatment of aortic valve disease. A sutureless bioprosthesis is implanted into aortic position in a transcatheter manner through peripheral artery (mostly the femoral artery) or through apex of the heart. TAVI is an effective alternative to standard surgical aortic valve replacement, particularly in elderly, inoperable and high-risk patients. The advantage of this procedure is the minimization of invasivity, the absence of extracorporeal circulation and cardiac arrest. It is however more expensive and has some technical limitations - the procedure cannot proceed in patients with very small or very big aortic anulus, infective endocarditis, aortic regurgitation, bicuspid aortic valve. Most frequent complications are paravalvular leakage and damage of heart conduction system. Recent guidelines recommend TAVI as a justified therapeutic modality in inoperable and high-risk group of patients. Nevertheless, results of the newest clinical trials show that it could have comparable outcomes to surgery even in lower-risk group.
TAVI represents a distinct approach in the treatment of aortic valve disease. A sutureless bioprosthesis is implanted into aortic position in a transcatheter manner through peripheral artery (mostly the femoral artery) or through apex of the heart. TAVI is an effective alternative to standard surgical aortic valve replacement, particularly in elderly, inoperable and high-risk patients. The advantage of this procedure is the minimization of invasivity, the absence of extracorporeal circulation and cardiac arrest. It is however more expensive and has some technical limitations - the procedure cannot proceed in patients with very small or very big aortic anulus, infective endocarditis, aortic regurgitation, bicuspid aortic valve. Most frequent complications are paravalvular leakage and damage of heart conduction system. Recent guidelines recommend TAVI as a justified therapeutic modality in inoperable and high-risk group of patients. Nevertheless, results of the newest clinical trials show that it could have comparable outcomes to surgery even in lower-risk group.
RESULTS
Early (30-day or in-hospital) mortality of isolated aortic valve replacement is 2.5% in average according to the most recent data. Surgical risk rises with age over 70, in urgent settings, re-do surgeries, in active endocarditis, pulmonary hypertension, heart failure, low ejection fraction of the left ventricle, diabetes, renal, pulmonary, neurological and atherosclerotic disease, and in concomitant cardiac surgery.
Long-term outcomes are very good. Median survival of patient after isolated aortic valve replacement is 12.8 years (surgery in 65-69 years of age), 9.2 years (70-79 years of age), 6.2 years (over 80 years of age). Risk of re-do surgery is very low in mechanical prostheses (2% in 25 years), in bioprostheses it is around 5% in 5 years, 10% in 10 years, but up to 30% in 15 years, strongly depending on the age of the patient (the older the patient, the slower the degeneration).
Resources:
- Vojáček J, Žáček P, Dominik J, et al. Aortální nedomykavost. Praha: Grada Publishing, a.s., 2016. ISBN: 978-80-247-5685-1.
- Češka R a kol. Interna. Praha: Triton, 2010. ISBN: 978-80-7387-423-0.
- Dominik J. Kardiochirurgie. Praha: Grada Publishing, 1998. ISBN: 80-7169-669-2.
- Baumgartner H, Falk V, Bax JJ, De Bonis M, Hamm Ch, Holm PJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. European Heart Journal 2018; 38: 2739-2791. doi:10.1093/eurheartj/ehx391
- Erbel R, Aboyans V, Boileau C, Bossone E, Di Barolomeo R, Eggebrecht H, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases. European Heart Journal 2014; 35: 2873-2926. doi:10.1093/eurheartj/ehu281.
- Dominik J, Žáček P. Chirurgie srdečních chlopní (... nejen pro kardiochirurgy). Praha: Grada Publishing, a.s., 2008. ISBN: 978-80-247-6386-6.
- Gofus J, Voborník M, Pojar M. Miniinvazivní přístupy v chirurgii aortální chlopně. Intervenční a akutní kardiologie 2018; 17(1): 24-28.
- Cohn LH, Adam DH. Cardiac Surgery in the Adult, Fifth Edition. New York: McGraw Hill Education, 2016. ISBN 978-0-07184487-1.
- Dominik J.: Kardiochirurgie. In.: Pafko P. et al.: Základy speciální chirurgie, Galén, 2008, s.173-195. ISBN 978-80-7262-402-7
- Dominik J. Chirurgická léčba chlopenních vad. In.: Vojáček J., Kettner J.: Klinická kardiologie, IV.vyd., Maxdorf, 2019, s.595-613. ISBN 978-80-7345-600-9
